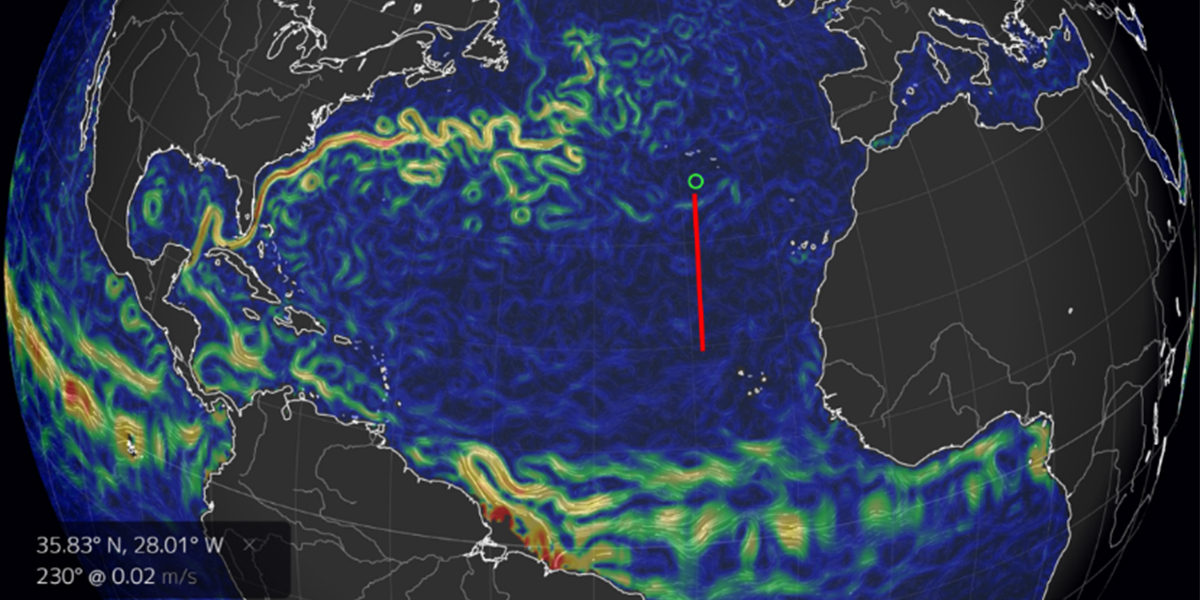
During earlier research, we demonstrated the importance of viruses as cause of mortality of the various different natural phytoplankton groups under summer conditions in the northeastern Atlantic Ocean (‘STRATIPHYT’ project; over a large geographical gradient from Canary Islands to Iceland). The Stratiphyt project also revealed that grazing dominated algal mortality north of 55˚N, while viral lysis (the death due to viral infection) dominated in the south. As these 2 types of mortality affect the flow of carbon and energy in substantially different ways, this switch in phytoplankton mortality has direct consequences for ecosystem productivity and element cycling. The underlying mechanism of this shift remain still largely unknown.
During a following cruise on the nortwestern side of the Atlantic Ocean (as part of the Dutch ‘NICO’ expedition) there was deeper mixing because we were there earlier in the year (spring), which resulted in reduced viral lysis rates. Grazing rates, however, were comparable to literature values recorded in spring and summer. These findings suggests that increased vertical mixing is disadvantageous for effective viral infection of phytoplankton populations (reduced chance to “bump” into the specific host). This escape of virally induced mortality would contribute to the build up of algal biomass at an early start of the productive season in spring.
Our current follow up Stratiphyt-21 cruise aims to prove that the degree of mixing and consequently the changes in light availability (deeper mixing results in reduced average light for the phytoplankton) impacts the degree of phytoplankton mortality by viruses. This information would provide the first experimental evidence for our hypothesis and also for a recently published theory related to the annual cycle of phytoplankton production.
8 March | Goodbye (for now)
By Corina Brussaard
We are currently sailing in the Channel and see land again. The weather is cold but the sea is nice and calm. We have packed all our equipment and stuff in 2 lab containers and are busy with getting the flow cytometers ready for packing. We are still measuring on one of them – we will close down that one tomorrow. It is secured in one of the lab containers and so we took advantage of that and keep measuring :)

The cruise has seen quite different weather and Anne, Robin and Tonke who sail for the first time on a scientific ship have been lucky with what they have encountered during just one cruise: dolphins, unbelievably flat sea with the sun and the full moon together in the sky. The full moon during the night shining over a flat sea was also a rare sight (even for the seafarers we were told), Sargassum with the associated animals (snail, shrimp, crabs) that look like Sargassum leaves, rocking ship from waves that come from the back, the side and the front, calm and rough(er) seas and blue skies and rain.
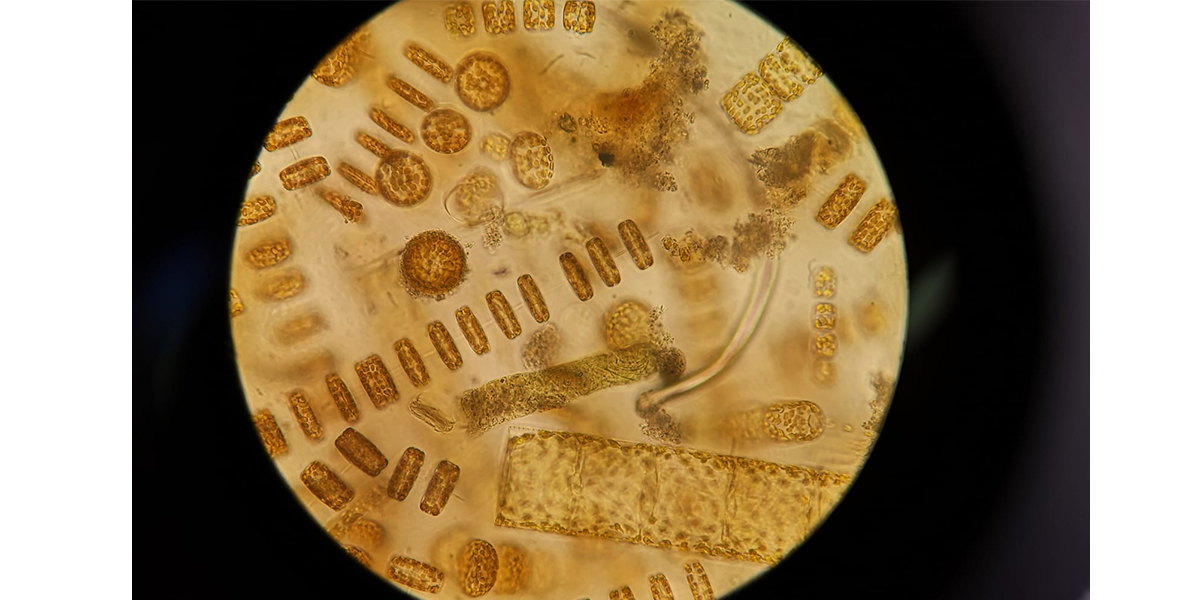
Today we concentrated seawater to look under the microscope. It is Spring and hence these temperate waters are teeming with life: diatoms, the algal cells that have an outer skeleton of silicate. Like glass they look and in them we see the pigmented chloroplasts where they collect the light and turn it, together with CO2, into sugars. Amazing how joyful one can get by just looking at them under magnification: the students are seeing them like this for the first time but personally I also get excited by these nice images of well growing algae.
The crew onboard and the scientific team have done a wonderful job and tomorrow we will arrive in the harbor of Texel, after which the scientific members go back to their homes and adapt back to COVID-19 regulations. The crew stays onboard for the next cruise to the North Sea. Now we all have our fingers crossed the weather will not be too bad for them, as the current prediction for coming Thursday and the days following is that there will be lots of wind!
Goodbye and I hope you enjoyed our blogs. Have a good day and see you next time we sail!
Cheers,
Corina Brussaard (Chief Scientist)
6 March | Almost back home
Looking back to our last sampling day it didn’t go as smooth and fast as we wanted because there was a change of weather. It was raining, more wind and the waves were a bit bigger. Even though the weather was different than the day before we still managed to get everything done. It was the first time we needed our rain gear. We were really lucky with the weather during this cruise; it made it much easier to do our lab work. Now it’s a lot of measuring and cleaning as described in the previous blog. Going back to Texel, it is getting colder and there is more often a bit of rain. We are wondering what type of the so called “Kanaalkoorts” (‘English Channel fever’) we will get when getting closer to home….
Today is a very special day for our second officer Kelly, it is her birthday. During coffee break she got a delicious chocolate cake made by the cook. We had early dinner as we started an extended pub quiz. The categories were all related to the sea and different people prepared questions for one of the 8 categories so we could all join the game. We prepared the prizes for the 4 teams, made from old filter cartridges we used during the cruise and which would otherwise be thrown away; one with ‘official’ Pelagia money and one with a beautiful group picture made on the back of the ship. It was time to start but first we enjoyed the pizza. Three rounds of mixed questions from all different categories raised the points on the leader board to an almost tied final score. It was time for the last round about sea animals, which was prepared as a describe and imitate game. This was lots of fun trying to imitate a starfish or a seahorse.
Tomorrow we will be still measuring and we will be packing. And enter the English Channel…

5 March | The last station, but the work Is far from over
By Corina Brussaard
The ‘last station’ sounds like it's all over, but this is far from the truth. Yes, we have stopped taking new water samples, but we are still measuring what is in the freezer. That just goes on all day, every day. There is also some administration to get done. We have made a lot of comparisons and I would like to get these in order as well. And, of course, there is the cruise report that needs to be written: what and where and how did we do which science?
It is very handy to have a simple overview of the samples and data for further processing, and we now extract this from the various logbooks. In this way, we can also check within the science team if something is not quite clear (such as a coding).
Today, I have been working on data obtained from the CTD (see earlier blog) that almost constantly measures the values for various parameters on the way to the deep and then up again. I want to make a table of these values during the closing of the water samplers, so that it is easy to look back later when we write up data for a scientific article.

In the meantime, all the stuff we used is being cleaned and dried. Rinse, rinse, rinse and repeat! After emptying and cleaning, the lab containers are set at a slightly higher temperature and the clean items are placed inside to dry.
Tomorrow and the day after everything will be further packed into the transport crates. Bur right now, it’s time to drink a glass of tea and relax a bit.
1 March | In between the ongoing work
By the scientific research team
We currently sail in Portugese waters that are super calm for this time of the year. This morning we saw two objects sticking out of the water, do the seabirds finally dare to land and rest a bit on the water? No! They are creatures that are no surprise to be found here due to their name: Portugese man o’ war. This colonial organism (no jellyfish) has a beautiful blue/purple “sail” that enables them to move by the wind.

The calm water allowed us to see into the deep and there were small jelly fish and tiny fish. Floating on these waters is Sargassum, long stretched patches. These seaweeds harbor Sargassum specific snails, fish, shrimp and small crabs that have the same color, and sometimes even shape, as the Sargassum. It’s about camouflage, after all.
There are many other things floating in between the Sargassum. It is like a net catching plastic and other trash. Now we are talking about plastics: every day a certain amount of water is filtered for algal pigment analysis (to be measured back at NIOZ) but on these filters (containing particles bigger then 0.7 micrometer) we also see small plastic particles and fibers.

This one day the ocean was very calm and this turned the surface of the water into a sort of mirror. This resulted in amazing reflections of the colors of the sky. Making it look like we sailed trough a cotton candy world!
Back to work now – working on the last station for this cruise. Thereafter we will continue counting samples that are in the freezer as we could not handle all the samples we generated per day.

27 February | Waves and sounds
By Corina Brussaard
The last days we had quite some wind and swell, which forced those who sailed for the first time to readjust to all these sudden movements. One of the consequences of such weather is that the table cloths are replaced by anti-slip material, to make sure all stays on the tables instead of sliding back and forth (and potentially tipping over on the floor). Furthermore, we check more often and more careful if all is tied up in the labs and containers. How often have I not seen things fall on the floor (including computers) or move & roll through the lab on the floor. Even the very heavy things may ultimately move and once they move, be sure they will make some damage!
The realization that this storm north of us did not make it as far south as predicted a week ago, makes me feel lucky. Considering this is an early spring cruise, we can only say: So far so good with the weather!
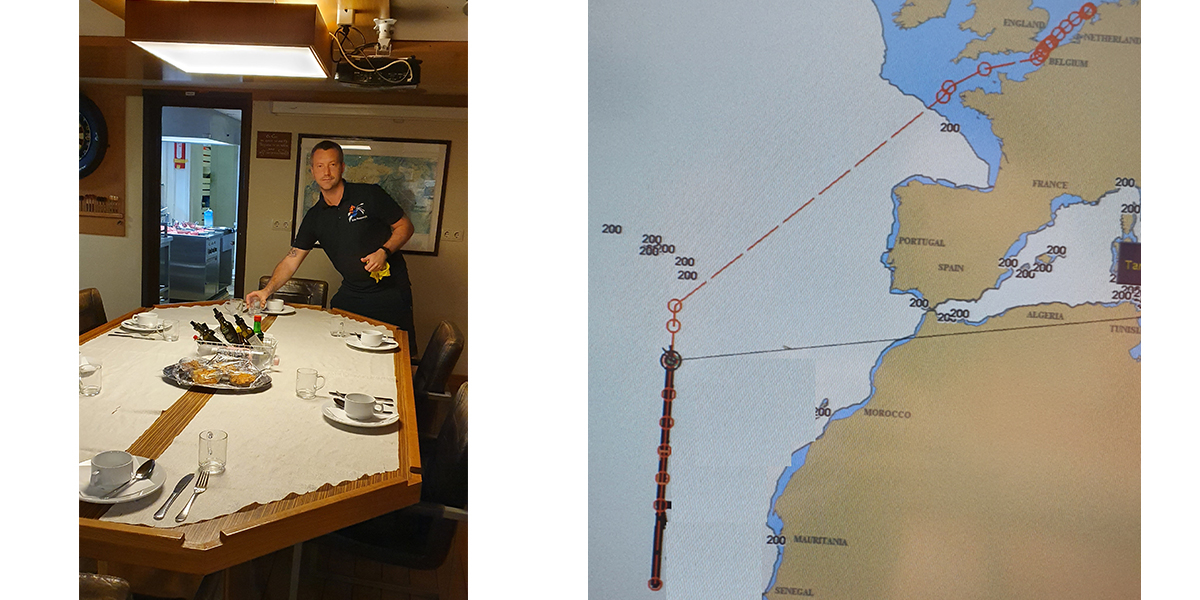
We know, we can explain, we understand, but still there is this strange and rich feeling again that is merely beautiful and somewhat amazing that this ship full of equipment and people stays afloat on all this water. She perseveres and along a straight line at 26 degrees West we sail north, stopping every 2 degrees (120 nautical miles) for water sampling and what more we do for science. While sailing we measure and measure and every so often take a few minutes to gaze over the wide ocean…
The ship hitting the waves the last days resulted in additional sounds directly made by the ship – the extra vibrations, creaking and sighing of the Pelagia at first alerts you but then after a while, it becomes all part of the background noise. Together with the air stream in each room from the ship’s climate control system, the engines’ humming, the noise our equipment makes, the occasional alarm from our equipment, and when we are on station also the sounds of the crane used to hoist specific equipment into the water. Still, these sounds are like an alarm clock for me – when one stands out or there is another sound, I look up. I even wake up just from the change in background sounds when the ship slows down in the early morning nearing the next station. When I am working and hear the crane start, I know I have to go as things are ready for scientific use. We are all working together like a well-oiled machine by now. The small but super-efficient scientific team together with the excellent support by the crew makes this cruise a pleasure to lead.

25 February | The ocean is always moving and so are we
By Kirsten Kooijman and Tonke Vogt
The ocean starts to move more due to increasing winds. During the night we cannot go on deck but have to access the small alley in front of our lab containers from inside the ship. But before yesterday, the ocean gave us quite a few internal waves, which are underwater waves! So cool to realize there is such motion under our feet. To keep up with the moving depth profile, we need to react! Tubes have been shuffled around and the experiment is adapted to the newest data. To act as soon as possible, a flexible mind is required. To obtain a flexible mind physical exercises can be key. And that’s what we do all day!

Starting with collecting empty jerrycans and bottles and containers, followed by filling them from the first CTD that went into the water around 6 AM. The, lifting jerrycans to the container, the working day is set! We prepare for the growth and loss assay, and empty containers while filling others with the produced filtrate. Lots of bottles are filled, small sampling tubes are rearranged on the bench…and BAM! The first set of fresh samples is ready to be measured.
Now quickly, the incubation bottles need to be transported to the on deck, flow-through incubators. This is the heaviest exercise of the day as we have 3 treatments with 24 bottles of 1 liter each (photo 1). Lifting weights is nothing compared to this exercise. With the four of us we manage to transport 144 bottles in less than 30 minutes on a moving ship and place them correctly in the incubators while preventing the lid of the incubator to take off due to the wind. The exercise is full of obstacles, like up and down the stairs, so safety needs to be considered.
To keep your mind fully functional, it’s important to give the brain some relaxation in the form of sports, doing a jigsaw puzzle or even knitting can be helpful (photo 2). For example, Kirsten misses her early morning bike ride to the TESO ferry and therefore goes biking in the gym at 6 am before the first CTD is out of the water! However, not everybody is so dedicated. Others prefer to do sportive activities in the form of table football. Some crew members also join the competition. After years of practice they are more experienced which occasionally frustrates the scientists. It is good to clear one’s mind before a deserved goodnight to start fresh and full of energy the next working day.

23 February | What we do after lunch
By Anne Mol
Tonke (one of the previous blogs) told you about what we do before the lunch on a normal sampling day, I will tell you what we do after lunch. After lunch its mainly measuring the phytoplankton from our growth and loss assays (the newly started as well as those after the 24-hour incubation). Furthermore, there is the cleaning, cleaning and some more cleaning. When the cleaning of bottles, containers and jerrycans is done we can chill a bit in the sun. Until the timer goes in alarm and we need to put another sample under the flow cytometer (every 10 minutes, 3 flow cytometers). In the meantime, we also write sampling tubes for the next day. This means we need to write on the tubes information about the sample, which station, date; these tubes will contain seawater samples that are fixed and frozen for analysis at a later point. When the writing is done I like to knit. I think everyone from the crew asked me at least once what I am making. For clarification, I am making a hat, and yes, I know it’s getting spring but days in spring can get really cold. Besides me, Tonke and Kirsten are also knitting, we form a little knitting club together. Interrupted by the timer alarm, we continue measuring on the flow cytometers until about nine in the evening.

Yesterday, we saw quite some plastic floating in the ocean while at station. The first thing we took out of the ocean was an old thrash bin. In it there was a trigger fish that we released back into the ocean. Then the crew managed to bring on board a huge entanglement of fishing nets and other plastics that got caught in it. For a lot of tiny crabs this was their temporarily home. Picking up the tiny crabs by hand was really difficult; with a dustpan Corina carefully swooped them up without damaging them. Many of these tiny crabs made it back to the sea (several hundreds were found on and under this plastic waste). And many different colours these small crabs had: ranging from blue to red to white-spotted on the back, to opaque shiny ones (such a pity we did not take photos; too busy saving them). One can only hope there wasn’t any fish swimming behind us and eating all the crabs we were trying to save.

22 February | Fishing for plastics

21 February | Daily life on board
By Robin Blonk
It feels like the rules of time do not apply to this ship. Days pass quickly and we are fully adapted to the life onboard. Our lab actions have become daily routine and we work efficiently as a team to solve problems that cross our way. Hassan, the cook, provides us with delicious meals and I am having a hard time trying not to eat too much. Today, he putted a lot of effort in preparing a BBQ that we enjoyed much with the calm sea and sunny weather. We decorated the deck with a palm-tree made of plastic bottles and lots of little flags. It was great to see the scientists and the crew sharing their stories and music preferences. Such a relaxing evening restores our energy that we need during this trip.

The next morning, we were surprised by a beautiful sunrise and we took a minute to enjoy the view. Now we’re heading north, we have more chance to see whales, so my eyes often lay over the ocean when I walk over the ship. The crew teaches us knots that we now often use to fix things. Some even take it a step further by making a huge hammock that I think will not fit at any place on the ship (picture). After sampling, in the late afternoon, we (the students) went to the front of the ship to relax a bit in the sun. I am jealous of the algae that are floating in these waters 24 hours a day.

19 February 2021 | Counting and characterizing phytoplankton cells
By Anna Noordeloos
During this cruise I am mainly working on the flow cytometer. This instrument is most common in clinical applications (hospitals) but it can also be used as a research instrument to count and characterize for example phytoplankton cells. The principle is simple. A seawater sample containing small-sized phytoplankton cells is passing a laser light. The light of the laser scattered by the individual cells is measured. At the same time there are sensors detecting different colors, either because of autofluorescence in the case of the phytoplankton cells (from the most dominant pigment in the algal cells) or because the cells were stained with a fluorescent dye for example. The molecules of the stain send out a bit of light. These signals are measured by the flow cytometer. Because the phytoplankton cells contain their own stain, namely the pigment Chlorophyll-a, we can count them one by one in the seawater samples we collect without further treatment.
To allow for optimal enumeration of the different phytoplankton populations in these waters, we count each sample of seawater for 10 minutes (equaling 1 mL of seawater). We take seawater samples down to 100 meters to be able to make a depth profile of the different algal populations in the water column. Today the maximum phytoplankton cells we counted was 200.000 per milliliter. Most of these cells are cyanobacteria with cell diameters of only around 1 micrometer (which is one thousandth of a millimeter). Still, just imagine! So many algal cells, together with about a million bacterial cells and 10 million viruses….YES, the ocean is full of microbial life….and we are there to study it, also in the weekend :)
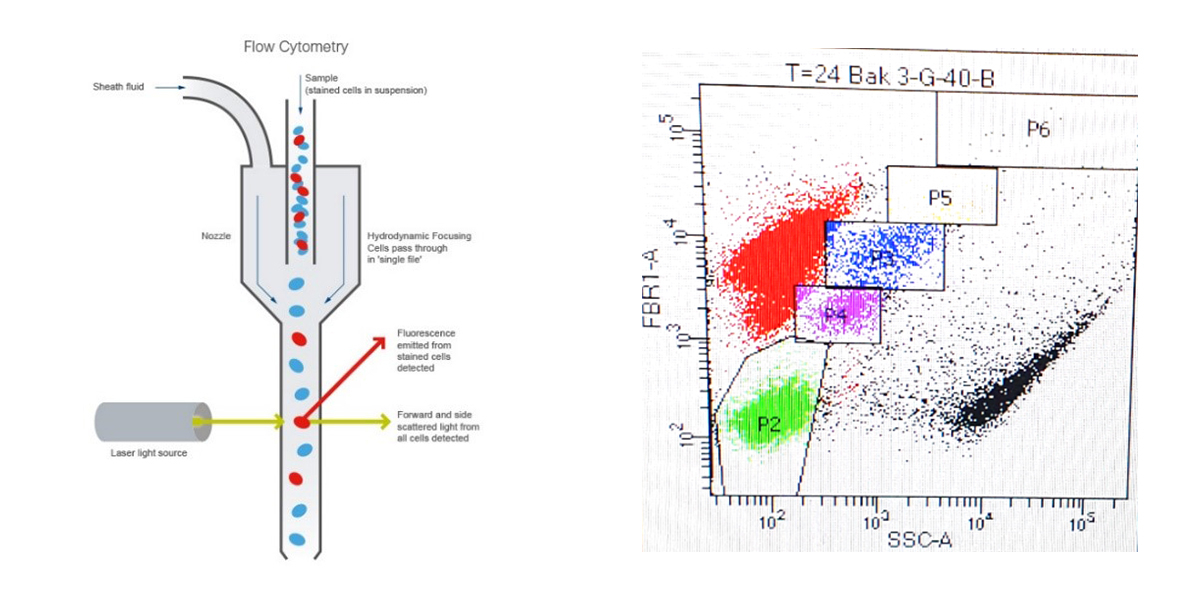
Explanatory notes to the figures:
Figure 1. Principle of a flow cytometer
Figure 2. The red Chlorophyll-a pigment of the phytoplankton (FBR1) cells versus the side scatter (SSC) signal displays different populations. For the photo we have roughly indicated them with various colors. For each of the different groups we determine the growth, viral lysis and grazing rates.
18 February 2021 | Start of the sampling
By Tonke Vogt
Today was the eight day that the RV Pelagia sailed us through the (thus far sunny) North Atlantic Ocean in southernly, towards Cape Verde. Four days ago, we arrived at the coordinates representing our first sample location, i.e. our first station. After the few days of preparation we were ready to shine with all equipment set up in the lab containers, having practised with the experimental setup and sampled a test CTD (to check the biological conditions). A CTD is a sampling device used to gain data about conductivity, temperature and depth (CTD) and at the same time it may contain a carrousel of bottles to take water samples at different depths (see picture here below).

Under supervision of Anna and Kirsten, we performed the first modified dilution assay which will provide us with phytoplankton growth and loss rates (both predation and due to viral infection). First, we need to filter the seawater to remove predators and with another type of filtration device we also remover the viruses. Then we make a dilution series from 100% to 20% original seawater, using the two different filtrates. The bottles are placed on a special wheel (rotating one round per minute) which again is placed in an on-deck flow incubator and controlled for a specific light condition. This way we can mimic the ocean environment and at the same time manipulate the light conditions. Samples for phytoplankton counting are taken at the start and after 24 hours of incubation. Using the difference in growth for the different dilutions we can estimate the growth and specific loss rates. We also sample for different other variables to help us obtain a good idea what is there and who is doing what.

At the morning of the first sample day, we were all looking excited how the first official CTD was placed back on the deck of the Pelagia filled with seawater from different depths of the ocean. Directly after that, we tapped the water out of the CTD bottles and started the filtration of the seawater. We tried to work as time efficient as possible, because it is preferable to incubate the samples early soon after it becomes light. Only working in a floating lab is slightly different compared to a working day at NIOZ. For example, while performing lab actions you need to keep standing upright when a (heavy) wave rocks the boat. It feels like your normal lab bench is moved into a ship attraction in an amusement park.
Because of this special work environment backpacks are used to safely transport the samples (and yourself) to the incubators which are located on the front deck on the other side of the ship. During our walk to the front of the ship we are looking over the endless (sunbathing) waves. Then, while fitting the bottles in the incubator we already smell the freshly made food. Luckily, after putting the samples in the incubator it was time for lunch.
18 February 2021 | Another day at the office ...

17 February 2021 |

15 February 2021 | Stratiphyt-21 pre-cruise preparations, part 2
Before a cruise can start there are a lot of logistics to deal with.
By Corina Brussaard
As Kirsten told earlier (in de blogpost below), there is a lot of packing to do before a scientific cruise actually takes place. Besides this essential activity, one also needs to think of where to sample and if diplomatic clearances are needed (in case you want to sample within the economic zone of the country involved), all the check and forms to fill in beforehand (medical check, sea survival training, personal detail form including information about potential allergies and info who to contact in case of emergency), and what shoe sizes your participating party has so the mandatory safety shoes/boots can be arranged. Then there are the material data sheets for the chemicals one brings on board, what category they fall, how much, where to store on board.
As cruise leader you have to prepare a station plan (where do we stop to collect samples), a sampling plan (what do we sample for and how much water or sediment is needed; by whom), and a time schedule to make sure all this can be done in the time given and corrected for the transit time (to get from A to B). The latter needs to keep in mind the potential weather conditions you can encounter – which depends on where you go and when.
Once on board you check your plans with the captain and discuss potential hick-ups (weather, timing, technical issues) and hopefully you have prepared a back-up plan as well

Due to COVID-19 we are restricted to 6 participants for the scientific party, so the scientific plans have to be adapted to this. Having done so, we originally planned to have 3 from NIOZ and 3 from the USA since this cruise project is a collaboration between Kristina Mojica (Univ Southern Mississippi, USA) and me (Corina Brussaard, NIOZ). Kristina arranged from her side the cargo and medical etc., but then, when they were flying in, they stranded halfway and after some intense discussion we decided they would go back home; the actual science time on board would have to be shortened to allow them to join after rescheduling their flights (with the risk to get stranded a second time), but also because they were advised by the travel company and embassy to keep in mind that borders may close which would mean they would not be able to fly back after the cruise ended. Of course, this was a serious disappointment for both her team and mine, not to mention the extra stress it gives on the science we wanted to do while on board.
With the help of our national marine facilities (NMF) officers Erica and Mildred, the quick check for safety and regulations by the head of NMF, the quick response from the 3 students in the lab I contacted whether they wanted to join (and said YES instantly, especially now they cannot perform lab work at the NIOZ due to the lockdown) and the help by Anna to guide them with other practicalities, we were able to arrange a new party of 3 in less than 4 days. Amazing!
Kristina and I have several intense discussions on what to focus on, what to skip if needed, how fastest we can get her model expertise for some of the on-board experimental planning passed over to me, etc. Once on board we had some additional challenges as you may have read in one of the first blogs; waiting for essential equipment that also got stuck but now in Madrid. Finally, it made it and off we went. Testing the most important equipment did not have the hoped-for result: the machine did not work properly. Now a few days later, Anna and I got it to work and we are also with this machine in business …. pff. Furthermore, the other instrumental challenges are working – also thanks to Mike, the ship’s electrician.

As you understand, this cruise needed quite some adaptation and rescheduling, including time planning and how much of the time goes to transit now we no longer stop at Las Palmas but go to Texel, and sampling station planning since it turns out some of the 10-year average expectations we do not hold up this year (see photo 1). The good thing with such a cruise is that everybody is willing to make it happen. It’s a pleasure to run this “show”. And it is nice to be called away from work to see dolphins in front of the ship! (see photo 2).
14 February 2021 | Stratiphyt-21 pre-cruise packing
Before a cruise can start there are a lot of logistics to deal with.
By Kirsten Kooijman
Last October 2020 we started making lists on what to bring, what we already had and what should be ordered. This year was different, COVID-19 is the reason why ordering stuff should be done as quickly as possible, you never know if and when it will come. So, the sooner it was ordered the better. But therefore we must check all the stocks to see what was already there. Different places in the NIOZ have been visited more than once to get everything together. Once we knew what to order we could only hope all would be delivered on time!
Still, we gained experience dealing with the COVID situation and we also organized a back-up plan. This includes for example making deals with other labs within NIOZ, swapping items, like ‘hey, this type of filter has a very long delivery time, can I have a box from you and as soon the item is delivered, you will get the new one? Everybody was willing to help in this way. And gladly, most of the ordered things were delivered on time.
Before loading there is the packing of all items that we need while being onboard. Protocols are the key here as they contain all the information about the methods used, not only ‘how to’ but also all what (materials, instruments and gadgets that are needed to make an experiment a success). Besides those we have also gained lists of items not to forget from all cruises which are always helpful. Thinking of spares, extra spares, more spares. It is better to have it and not use then to not have it and need it. When you are leaving shores there is no way back and you have to deal with the stuff that you have packed.
Thus, checking, double checking and then pack it all as efficiently as possible. This time we could load on Texel but often we have to send the equipment and consumables abroad and then we need to avoid having unused spots in the boxes. You can compare it with a matrouska doll, we put boxes in a box, and in that box you find small items like for example plastic tubes, timers etc. We pack most of our stuff in sturdy aluminum and plastic boxes with lids, small and big ones. Heavy stuff goes in small ones (to prevent too heavy lifting) and light items like plastic consumables are going into the big ones. Not only the actual weight and distribution of the weight is important but also the use of space. All that goes in the boxes is registered to prepare so called Track & Trace files and in case it is send of we have to prepare cargo bills so custom forms can be generated.
This list became my brain-backup, after filling 30 boxes my memory storage was full...

We loaded all the boxes in two of the lab containers that we need during our cruises. Those containers and much more, for example our deck incubators but also equipment of other cruises before and after ours, were all hoisted onboard the first week of January. We said goodbye to the containers and the ship: see you in February!
Now we are on board we unpack all to prepare dedicated labs (lab containers and ship’s labs): each for their own specific use. If necessary we first clean the lab containers as our microbiology work needs clean surfaces and floors.
The last days all equipment have been tested, the students joining have been training for the real thing. Today we practice with some of equipment in the water. Tomorrow we start for rea – the first station. Then we will be busy collecting data!

12 February 2021 | The next days are going to be wet
By Robin Blonk
Today the sun is still shining and the view is stunning; we are completely surrounded by the ocean and we were even greeted by a few dolphins. In the morning, a safety test was started by a loud alarm that tested us on how to act when we have to abandon the ship. Later, we practised the lab experiments while adapting to this new floating environment. Walking around with heavy bags of water samples requires a good balance. We now understand that the next few days are going to be wet.

11 February 2021 | Finally, departure!
By Robin Blonk
Finally, departure! Two days have passed since we set foot on the Pelagia. Our minds are focused on the package that can now arrive any minute. Our time is well spent by preparing the container labs where we tighten everything to prevent movement when we sail. I still have no idea how the wiggling is going to feel and the crew members are betting on what student is the first to get seasick. At the back of the ship is a great spot where we often settle to read the protocols and enjoy the sun. Looking down in the water between the ship and the dock we can see colourful fish eating mussels from the rocks. I wonder what creatures we will see in the coming days. The evening had room for a game of Qwixx that was interrupted by the delivery of our package! The crew immediately gathered on the deck to make the ship ready for departure. Soon the bridge was lifted and off we went. It was already late and while the air cooled down we saw the lights of Las Palmas slowly fade over the horizon.
10 February 2021 | Traveling to Las Palmas and waiting
by Anne Mol
When I was asked if I would like to join a cruise, of course I said yes immediately. But I had less than a week to take care of things. Normally this does not happen but because of COVID-19 things are different. After a week of organizing, sea-survival, COVID PCR-test, medical certificate, flights etc. a new problem arose soon before our departure. For the first time in 10 years there was a snowstorm going over the Netherlands. Just when we were about to leave. To be sure everybody would arrive at the airport in time we stayed in hotels close to Schiphol. The next morning, we checked in and all seemed fine. It was really quiet at Schiphol, we were the only flight going from our terminal. We waited for the boarding of the plane, but then the announcements came: issues with the snow water tank truck, fuel truck stuck, cabin manager still not there…. We just needed to wait and hope our flight would still be able to take off to Las Palmas. YES, after about 3 hours we could board. Waiting for the de-icing of the plane (see picture), taxiing over a white Schiphol and off we were. Landing in Las Palmas, it was nothing like the snow and ice we left in the Netherlands. We were picked up by a bus that drove us to the hotel. Enjoyed the delayed lunch which was arranged in the hotel. To stay in our “bubble” of no-COVID, we had to stay in the hotel. But we had a great view!

Next morning, we were picked up to go to RV Pelagia. On board we got each our own cabin and unpacked our stuff. I was very excited to be on Pelagia. We helped with carrying the food supplies onboard, and we already cleaned the lab containers. We were ready to leave. However, there was one very important thing still missing: the equipment from the USA partner (who could not make it due to travel logistic; that is why 2 other students and I could join the cruise at such last minute). Lots of uncertainties at the end of the courier; where was one of the 4 boxes etc etc. Stress at our end as it was completely unclear why the other 3 boxes that were in Madrid already for more than a week could not be send to Las Palmas. Corina (Chief scientist) was busy handling this. We just keep unpacking lab material while waiting. In the evening we played table football but some of the crew members were too good for me, I hope to be better at the end of the cruise.

Next morning, I woke up and opened my curtains and saw the harbour (picture). Corina could tell during breakfast that there was hope the missing boxes would make it to Las Palmas before the Corina’s deadline of 10 Feb at noon. After breakfast we got the task to paint some numbers on our deck incubators so we would know easily what treatment will be in which incubator. After lunch Corina, Anna and Kirsten explained us what exactly we are going to do and what the scientific interest behind all this is. Cor showed us an amazing spot on top of the bridge where you can chill or if you need to have some time for yourself. We really enjoy the sun so far and I look forward to start sailing to our first sampling location (station). We ended our day by finishing a beautiful jigsaw puzzle about Texel.

